Michael Faraday Inventions and Discoveries
He developed the first dynamo in the form of a copper disk rotated between the poles of a permanent magnet, the precursor of modern dynamos and generators. From his discovery of electromagnetic induction (1831) stemmed a vast development of electrical machinery for industry. In 1825, Faraday discovered the compound benzene. In addition to other contributions he did research on electrolysis, formulating its two laws. He laid the foundations of the classical field theory, later was fully developed by J. C. Maxwell. In his work on static electricity, Faraday demonstrated that the charge only resided on the exterior of a charged conductor, and exterior charge had no influence on anything enclosed within a conductor. This is because the exterior charges redistribute such that the interior fields due to them cancel (the Faraday cage principle). To demonstrate his ideas, Faraday built in 1836 a room (the Faraday cage), coated with metal foil, and allowed high-voltage discharges from an electrostatic generator to strike the outside of the room. He used an electroscope to show that there was no excess electric charge on the inside of the room's walls and volume. How does a Faraday Cage work?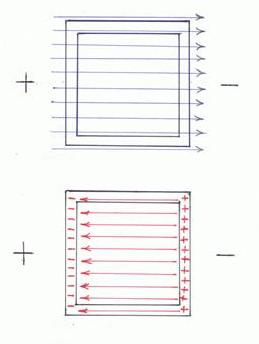 In the upper picture an external electrical field is created (blue lines) in the direction from plus to minus. Immediately after this field is applied, the electrical charges inside the cage walls are rearranged in the way depicted in the lower image since opposite electrical charges attract each other. As a result, is created another electrical field (red lines) between the cage walls in the opposite direction to the original field and with equal magnitude, and then, these two fields are cancelling each other and the electrical charge inside the cage would remain neutral. Another explanation for the Faraday cage effect can be derived from Gauss's law that states that if there is no charge in a closed surface then the net flow of electric field from the surface must be zero. Faraday cage is sometimes referred to as “Faraday's Ice Pail” because Faraday also used for his experiments a metal ice bucket as the receiver for the electroscope. Faraday Cage ApplicationsTake notice that Faraday cages also shield the interior from external electromagnetic radiation, besides from electrical fields, if the conductor or wall is thick enough and any holes are significantly smaller than the radiation's wavelength. This shielding effect is used to protect electronic equipment from lightning strikes and other electrostatic discharges, for RF (radio frequency) shielding and for shielded telecommunication cables.Practical Faraday cages can be made of a conducting mesh instead of a solid conductor. However, this reduces the cage's effectiveness as an RF (Radio Frequency) shield. Some real-world structures, such as automobiles, behave approximately like a Faraday cage. For example: If lightning hits near a car, it does not affect the people sitting in the car. Some traditional architectural materials act as Faraday shields in practice. These include plaster with wire mesh, and rebar concrete. These will impact the use of cordless phones and wireless networks inside buildings and houses. The shield of a screened cable, such as the coax used for cable television, protects the internal conductors from electrical noise. A shopping bag lined with aluminum foil acts as a Faraday cage. It is often used by shoplifters to steal RFID tagged items (don't try it!). For heavy duty tasks is recommended to ground the shield in order to receive better results. Demonstrate the Faraday Cage PrincipleFirst, build a simple electroscope according to the following image and instructions. 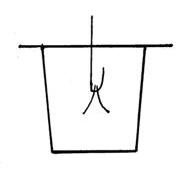 Prepare a hook from a paperclip or a wire. Cut two small aluminum foil strips (leaves) (1 x 3 cm) and hang them, not opened up, on the hook, as shown in the picture. Prepare a cardboard square, big enough to cover the glass or jar, and push the hook through it and place them on the glass as shown in the picture. Test your electroscope by touching the hook terminal with a charged straw or comb - the aluminum leaves will diverge since same electrical charge reaches the leaves and they will repel. Now, touch the terminal with your finger and the aluminum foil strips will be closed together again because the terminal is grounded and the charge is transferred through the human body into the earth and the leaves are now neutral. After your electroscope is ready:
More quick experiments:
Faraday’s "Ice Pail" Experiment - The Open Door Web Site Faraday's Ice Pail Experiment - Catharine H. Colwell Faraday's Ice Pail - J. B. Calvert Easy Electrostatics Experiments - Michel Maussion, University of Nantes, France Faraday cage - Holland Shielding Systems Faraday Cages in Health Care EMI Shielding EMC/RFI Solutions - Holland Shielding Systems Faraday Cages, TEMPEST equipment - Holland Shielding Systems Faraday's Cage - MIT Physics Demo Science fair projects and experiments: Static Electricity Science Fair Projects and Experiments Michael Faraday BooksMichael Faraday biographies and general resources: Michael Faraday - The Royal Institution Michael Faraday - Science Museum Michael Faraday - MacTutor Michael Faraday - BBC Michael Faraday - Eric W. Weisstein |
| Home | Fair Projects | Experiments |
| Scientists & Inventors | Science Jokes | Warning! |





webmaster@julianTrubin.com
